
Insulin resistance is the cause of both prediabetes and type 2 diabetes – a condition that desensitizes both your muscle and liver to the action of insulin. Living with insulin resistance increases your biological need for insulin, which forces the beta cells in your pancreas to overproduce insulin.
Over time, many people with prediabetes and type 2 diabetes end up damaging beta cell function, resulting in insufficient insulin production (1). The main cause of this beta cell dysfunction is the accumulation of fat inside your muscle and liver (2–7).
When your liver and muscle become insulin resistant, there are two solutions:
- Decrease your level of insulin resistance using low-fat, plant-based, whole-food nutrition, or
- Start injecting insulin
The primary function of the beta cells in your pancreas is to store and release insulin. Insulin is the hormone that helps your body utilize glucose – the building block of carbohydrates and your body’s primary source of energy.
When beta cells fail to work properly, your body cannot uptake glucose efficiently, trapping large amounts of glucose in your blood – a metabolic state called hyperglycemia (or commonly referred to as “high blood sugar”).
In a study published in the journal Nutrients last month, researchers asked whether a low-fat, plant-based dietary intervention improve beta cell function.
They chose to study a plant-based diet because of ample evidence showing that reducing animal product intake (high in nutrients known to promote insulin resistance, including fat, protein, and heme iron) can not only improve diabetes health, but reverse diabetes altogether (8–14).
The Study Design
The participants in the study were men and women between the ages of 25-75 years. Each participant was overweight (BMI between 28-40) but was not currently living with diabetes.
Half of the participants were in the control group, in which they made no dietary changes and asked to continue their normal eating habits.
The other half of the participants were asked to eat a low-fat, plant-based, whole-food diet, which consisted of approximately 75% of calories from carbohydrates, 15% of calories from protein, and 10% of calories from fat.
Participants in this group ate legumes, whole grains, vegetables, and fruits, and did not restrict portion size or calorie intake. They were asked to avoid eating any animal products and added fats (namely vegetable oils).
Participants’ beta cell function and insulin sensitivity were measured at baseline and at the end of the study (16 weeks).
Results
Over the course of 16 weeks, those in the intervention group who ate a low-fat plant-based diet experienced the following metabolic changes:
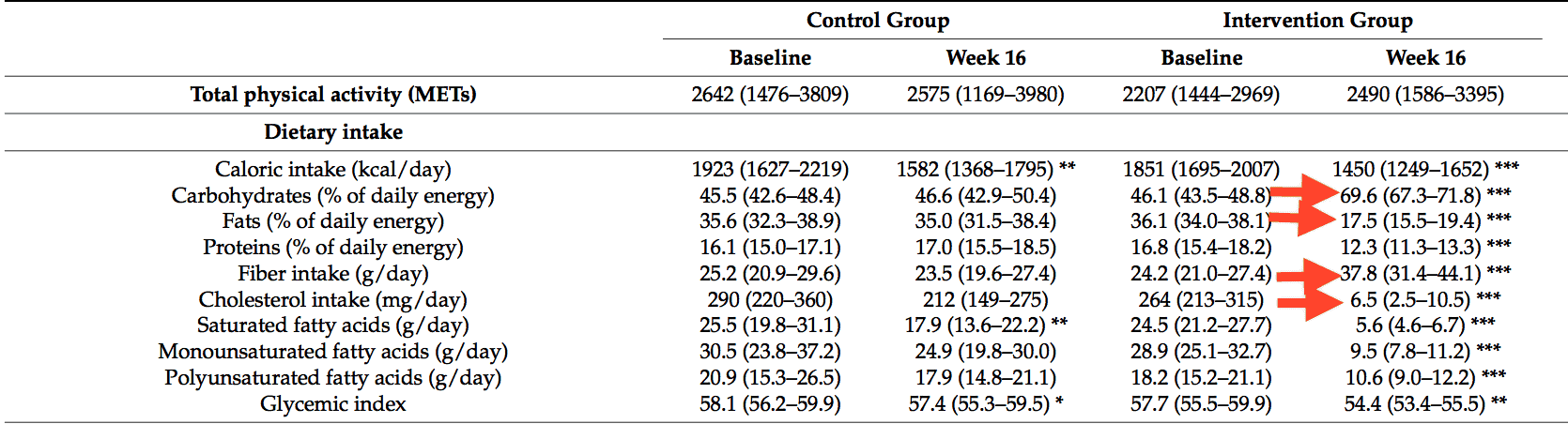
- They increased their intake of fiber by an average of 14 grams per day
- They ate an average of 50% more calories from carbohydrates per day
- They reduced their cholesterol intake by an average of 258mg per day
- They consumed an average of 52% less fat per day
Both groups experienced a decrease in lean muscle mass (about 1kg, or 2.2 pounds), but while the control group experienced an increase in fat mass, the plant-based group lost an average of 9.5 pounds (4.3kg) of fat.
In addition, the plant-based group lost an average of 13.7 cubic inches (224 cubic centimeters) of visceral fat, while the control group gained an average of 1.5 cubic inches (24 cubic centimeters) of visceral fat.
Let’s put that in perspective, because it is pretty impressive but probably hard to conceptualize at first.
Imagine a cube that is 1 inch x 1 inch x 1 inch. Now imagine 14 cubes this size, all connected together. Next, imagine that all of those cubes are made of body fat. That’s how much visceral fat each person in the plant-based intervention group lost on average.
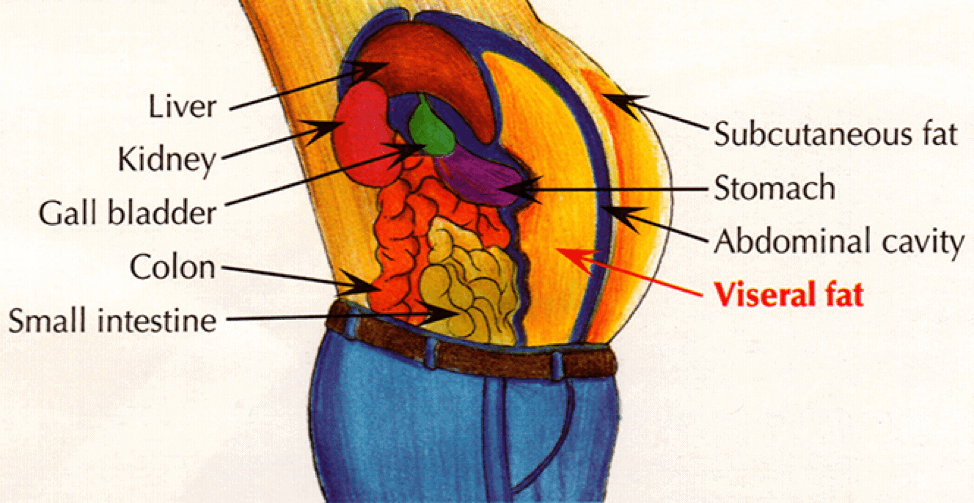
Losing visceral fat is particularly important for maximizing your diabetes health. Visceral fat is located in your abdomen and surrounds your liver, stomach, and intestines.
Visceral fat is known to increase blood pressure and insulin resistance, and increase your risk for chronic diseases like heart disease, type 2 diabetes, various cancers, and Alzheimer’s disease (15,16).
Subcutaneous fat on the other hand, lies directly beneath your skin and has a weaker correlation with chronic disease. See the image below to understand the difference between the two fat depots.
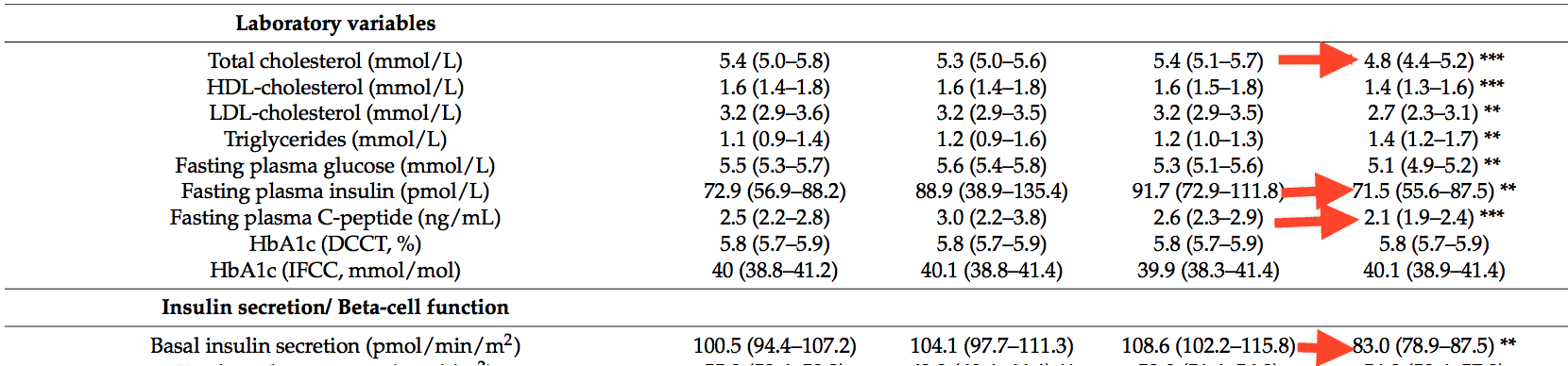
The plant-based group also experienced a significant reduction in total cholesterol, from a starting average of 209 mg/dL (5.4 mmol/L) to an average of 186 mg/dL (4.8 mmol/L).
What About Changes in Insulin Secretion and Beta Cell Function?
The 16-week intervention led to clear increases in insulin secretion and beta cell function.
Study participants in the plant-based intervention group experienced the following, on average:
- A 23.5% decrease in basal insulin secretion
- A 19% decrease in fasting c-peptide
- A 22% decrease in fasting insulin
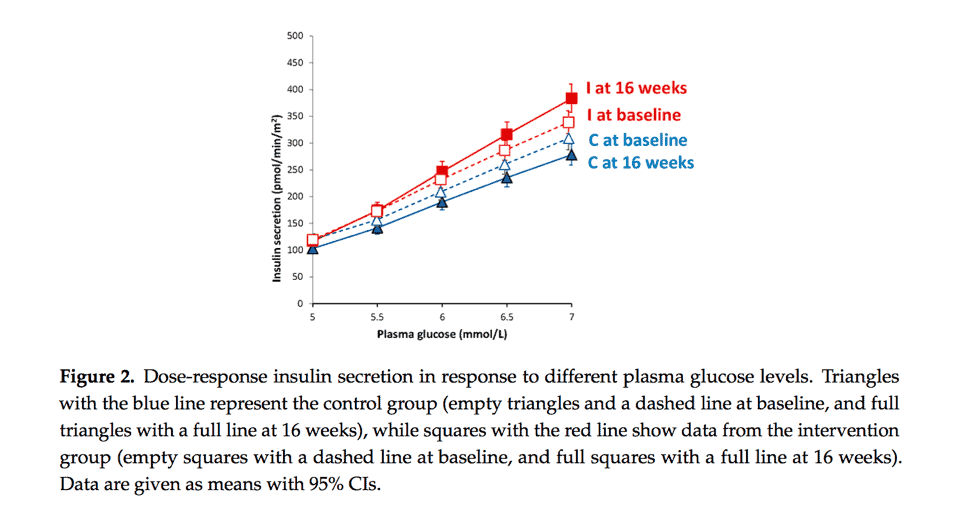
Participants on the plant-based diet experienced an increase in insulin sensitivity as well, though it was not statistically significant. However, they did experience a 24% decrease in insulin resistance, as measured by HOMA-IR.
What This Study Means and Why It Matters
The dietary intervention in this study is similar to what we recommend here at Mastering Diabetes – a diet rich in fruits, vegetables, legumes, and whole grains, devoid of animal products and high-fat foods.
This study demonstrated that eating a low-fat, plant-based, whole-food lifestyle reduces insulin resistance, reduces visceral adiposity, and increases beta cell function.
The two main pathophysiological mechanisms of diabetes (beta cell function and insulin sensitivity) improved at the same time from a single dietary intervention.
These improvements were the result of lower fat intake, lower intake of protein from animal sources, and increased intake of unrefined carbohydrates.
Furthermore, it is important to remember that study participants were not living with diabetes at the beginning of the study. If they had been, we predict that they might have experienced bigger improvements in beta cell function.
More Support Against Low-Carbohydrate Diets
Despite continued popularity with low-carbohydrate and ketogenic diets in the diabetes community, research repeatedly shows that low-carbohydrate diets are not the best solution.
Low-carbohydrate diets are inherently high-fat and high-protein diets, which have been proven to promote insulin resistance and increase your risk of other chronic diseases.
We recently wrote a comprehensive blog post about common misconceptions of ketogenic diets in which we discuss the drawbacks of reducing your carbohydrate intake to manage diabetes.
If you are using a low-carbohydrate diet to control your blood glucose, you may experience flatline blood glucose, however this occurs at the expense of increased insulin resistance.
Reduced blood glucose tricks you into believing that you are reversing insulin resistance, when in reality your risk for all chronic disease is increasing.
A low-fat diet, plant-based, whole-food diet on the other hand, is the key to reversing insulin resistance.
As shown in this recent study, a low-fat, plant-based, whole-food diet may improve your ability to utilize glucose.
People who adopt a low-fat, plant-based, whole-food diet are able to fully reverse prediabletes and type 2 diabetes, lose weight, reduce cholesterol, triglycerides, and blood pressure, and maintain these health improvements over time.
This study is yet another piece of evidence that when it comes to diabetes nutrition, a low-fat, plant-based, whole-food diet is an incredibly effective technique for improving beta cell function and maximizing diabetes health.
Lower Your A1c and Get to Your Ideal Body Weight ... Guaranteed

Your results are guaranteed. Join more than 10,000 ecstatic members today
Personalized coaching puts you in immediate control of your diabetes health, helps you gain energy, improves your quality of life, and reduces or eliminates your meds.

